Studying the mechanical properties Immunoglobulin Binding Protein (BiP) by Optical Tweezers and Nanorheology
Prof. Dr. Christian A.M. Wilson,
Laboratorio de Bioquímica, Facultad de Ciencias Químicas y Farmacéuticas, Universidad de Chile
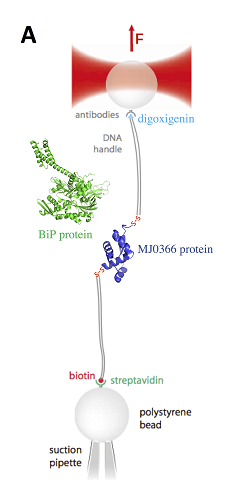
The effect of force on protein structure and associated changes of protein function is a subject of current intensive research. Optical tweezers are a useful research tool for applying forces to single proteins and measuring the affinity between substrates. The development of this technique by Arthur Ashkin was recently awarded the 2018 nobel prize in physics for its applications in biological systems. Some single molecule techniques that exert force on the protein are not able to measure small changes in distance at subnanometer resolution at low forces (below 1 to 5 pN), so it is difficult to correlate the elastic properties of the folded protein with ligand binding sometimes. Recently, a new technique called nanorheology allows measurement of elasticity in folded proteins. Nanorheology is a technique that exploits sub-Angstrom resolution to study the mechanical properties of the folded state of proteins by applying low force with 20 nanometer gold nanoparticles to the proteins in bulk. This viscoelastic transition is a universal mechanical property of the folded state, and it is relevant for the large conformational changes, which often accompany substrate binding in proteins. In this presentation I will show our recent results about the mechanical properties of chaperone BiP. FONDECYT-1181361, PCIPII20150073.
Viernes 19 de Octubre, 16:00, Sala Luciano Laroze E-300
Avenida España 1680, Valparaíso, Chile
Ver PDF